Our Passion for Research
At the Di Polo Lab, we explore how neurons in the visual system survive, communicate, and regenerate. Our research bridges cellular neuroscience and translational vision science, uncovering the metabolic, vascular, and mitochondrial processes that determine whether retinal neurons remain healthy or succumb to disease. By integrating state-of-the-art imaging, molecular therapies, and preclinical models, we seek to transform fundamental discoveries into therapies that protect and restore sight.
Theme 1 — Metabolic and Trophic Regulation of Neuronal Integrity
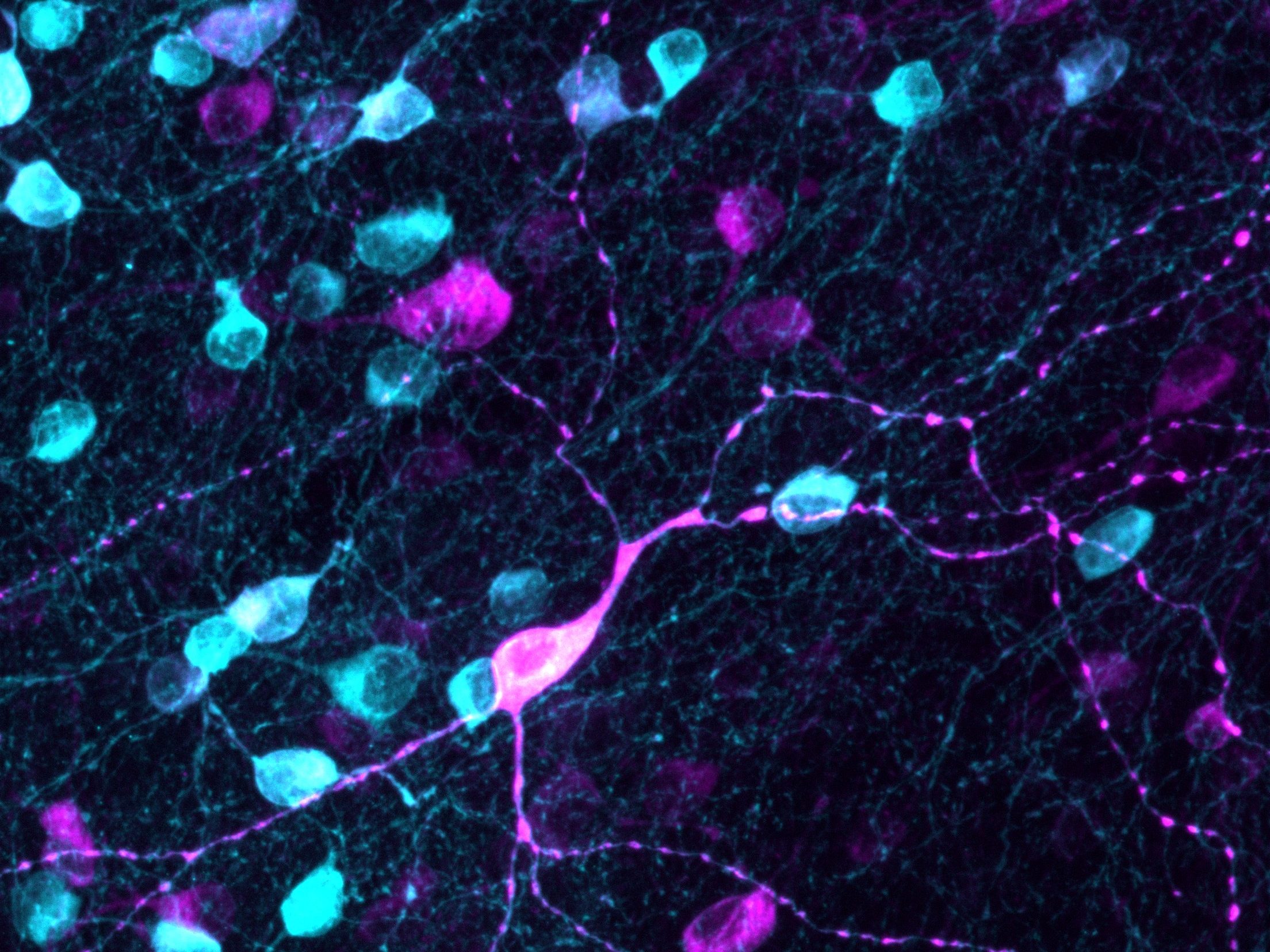
Our work has revealed that early deficits in energy supply and nourishing support are key drivers of retinal ganglion cell dysfunction in glaucoma. One of our major discoveries is that insulin, a naturally occurring hormone and growth factor, can stimulate the repair of damaged neuronal connections and restore visual function. By targeting these metabolic vulnerabilities, we have identified regenerative strategies that enhance neuronal survival and resilience. These findings highlight the remarkable capacity of neurons to repair themselves when provided with the right metabolic and trophic cues.
Theme 2 — Neurovascular Communication and Blood Flow Regulation
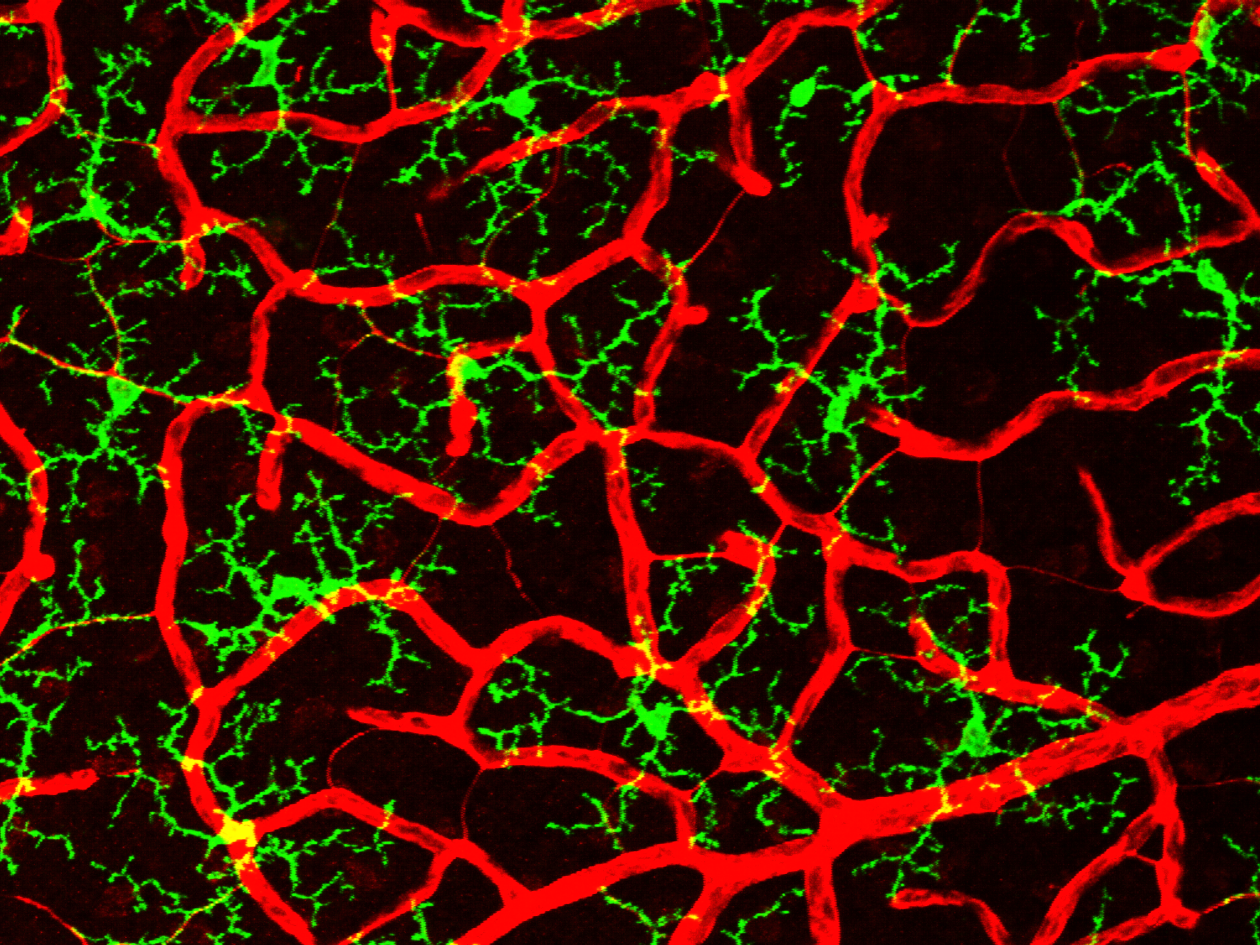
Using advanced live imaging and functional analyses, our team discovered inter-pericyte tunneling nanotubes (IPTNTs), microscopic bridges that allow small cells on blood vessels, called pericytes, to communicate and coordinate blood flow. Preserving these tiny structures restores the retina’s ability to adjust blood flow in response to light, maintaining healthy neuron–vessel communication.
We are also investigating how the diverse cells that make up the neurovascular unit — including glial cells, endothelial cells, pericytes, and neurons — interact to regulate local blood flow and how their miscommunication contributes to vascular dysregulation in glaucoma. Together, these studies are reshaping our understanding of how the retina maintains its delicate balance between neural activity and vascular supply in health and disease.
Theme 3 — Mitochondrial Function, Organelle Stress, and Axonal Transport
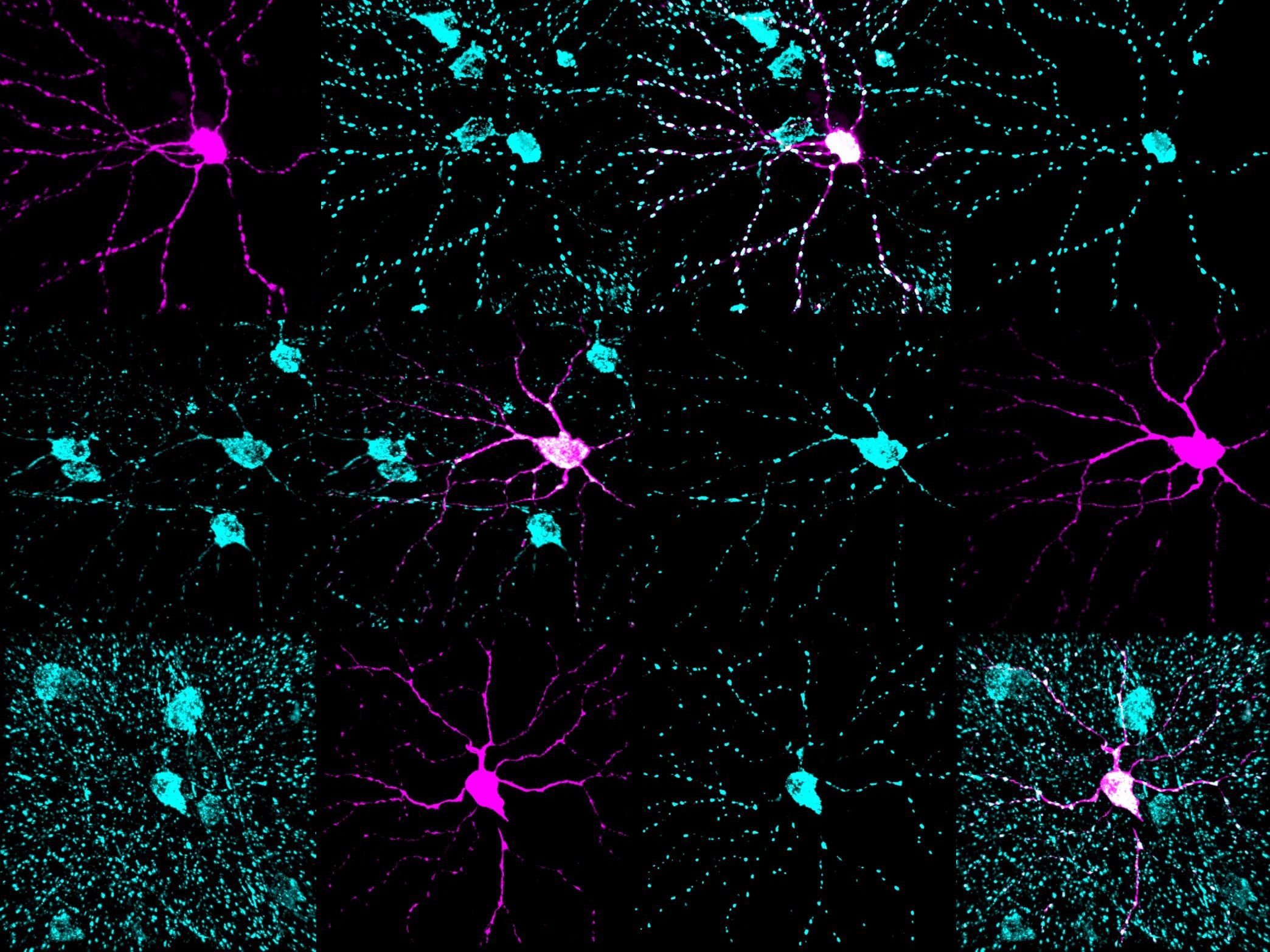
Mitochondria are the cell’s energy powerhouses, and their movement within neurons is essential for keeping vision cells healthy. Our studies show that when mitochondrial transport is impaired — as in glaucoma — energy levels decline and neurons become more vulnerable to degeneration. By using targeted gene therapies to restore this mitochondrial health and transport, we can boost energy production, protect axons, and open new avenues for vision preservation.